Research Article
Assessment of Growth and Survival Rates of African Catfish (Clarias gariepinus BURCHELL 1822) Fry Fed On Soybean Milk-Based Diets 
 Author
Author  Correspondence author
Correspondence author
International Journal of Aquaculture, 2016, Vol. 6, No. 7 doi: 10.5376/ija.2016.06.0007
Received: 11 Sep., 2016 Accepted: 28 Oct., 2016 Published: 23 Nov., 2016
Chelewani A.P., Kassam D., and Chiwanda V.J.M., 2016, Assessment of growth and survival rates of African catfish (Clarias gariepinus Burchell 1822) fry fed on soybean milk-based diets, International Journal of Aquaculture, 6(7): 1-10 (doi: 10.5376/ija.2016.06.0007)
African catfish (Clarias gariepinus) has higher potential yield than tilapia species farmed in Malawi. However, production is hindered by shortage of fingerlings due to mass mortality at fry stage attributed to poor feeding. A 42-day feeding trial was conducted at Bunda fish farm (14°10'25.6"S 33°48'21.6"E) of the Lilongwe University of Agriculture and Natural Resources, Lilongwe, Malawi to assess growth and survival rates of African catfish fry fed on soymilk based diets. In this experiment, four thousand five hundred fry (average weight; 0.0288 g ±0.0068) were fed on two soymilk based diets and an egg yolk based control diet, each replicated thrice. Significantly higher percent weight gain and specific growth rate was observed in fry fed on egg yolk based diets and diets based on soymilk from roasted soybeans compared to fry fed on diets based on soymilk prepared from raw beans. No significant differences were observed between catfish fry fed on egg yolk based diets and catfish fry fed on diets based on soymilk from roasted soybeans. Survival percentage was highest (42.46±5.93558) in fry fed on a diet based on soymilk from roasted soybeans. This was found to be significantly higher (P=0.05) than survival percentage of fry fed on egg yolk based diet (18.33±3.0118). No significant differences in survival rate (%) were recorded in fry fed on diets based on soymilk from roasted soybeans and soymilk from raw soybeans (36.93±5.42873). The results demonstrate that diets based on soymilk from roasted soybeans can be used to improve survival of African catfish fry and improve fingerling production, the biggest bottleneck in African catfish production in Malawi.
Background
In Malawi, per capita supply of fish from capture fisheries in recent years has steadily increased due to the abundance of the small cyprinid Usipa (Engraulicypris sardella) which constitutes 70% of the total catch from Lake Malawi (Njaya, 2015). In spite of this, fish supply from capture fisheries continues to fall short of the demand of one of the fastest growing populations in the SADC region. This has resulted in decline in fish per capita consumption in the country from 14 kg/person in the 1970s to about 5.6 kg/person (USAID, 2015). The development is of particular nutritional concern considering that fish provides over 40% of total dietary protein and 72% of the dietary animal protein in Malawi (Ecker and Qaim, 2011). Clearly, the potential of aquaculture as a complementary source of fish has to be further developed if fish is to remain an important source of food, nutritional and economic security for many Malawians.
Current combined production from small scale and large scale fish farming accounts for around 2% of the country’s total fish production, about 3 500 tones (Njaya, 2015). Increased fish production from aquaculture would require, among others, the selection of suitable species for intensive production. Currently, Malawian aquaculture is dominated by the production of three tilapia species, namely; Oreochromis shiranus, Oreochromis karongae and Tilapia rendalli. Among them, the three species account for over 90% of production with Clarias gariepinus contributing to around 5% of total production (ADiM, 2005; FAO, 2006; Iita, 2013). The major problem with the production of the three tilapiine species is slow growth rate leading to stunting where females tend to become sexually mature and reproduce early at a small size (M’balaka et al., 2013).
In contrast, the African catfish has received recognition as the most robust species for Aquaculture in Africa (de Graaf and Jensen, 1996; Nagoli and Balasubramaniam, 2008). The African catfish is an excellent species for aquaculture as it is omnivorous, grows fast; able to reach 800 g in seven months, tolerates relatively poor water quality and can successfully be stocked at very high densities (Borade et al., 2001; Rad et al., 2003; Hecht, 2013). In addition, the yields of African catfish from ponds can be as much as 2.5 times higher than those of tilapia (Abdelhamid et al., 2010).
The species occurs naturally throughout the country, inhabiting calm waters of lakes, rivers, swamps and flood plains, and thus is ecologically safe to farm (Nagoli and Balasubramaniam, 2008). The widespread culture of the species would not only prove more profitable among local fish farmers but also ease the need to lift the ban on the introduction of exotic species that have better aquaculture output than Oreochromis shiranus and Tilapia rendalli, such Nile tilapia (Oreochromis niloticus). Thus, the African catfish is both a profitable and sustainable alternative to the three tilapia species commonly cultured in the country.
There are some challenges facing widespread Africa catfish culture that help in explaining the country’s marginal production of the species despite its superiority to the tilapia species. Inability of African catfish (Clarias gariepinus) to breed naturally in captivity and high mortality rates during larvae and fry stages have long been recognized as the major hindrances to wide spread production of the species (Goos and Richter, 1995). The introduction of synthetic hormones for induced propagation, albeit expensive, has tremendously improved seed production. However, achieving satisfactory survival rates remains an ever present challenge. To put this into perspective, Iita (2013) reported that survival rates of less than 10% are very common among private hatchery owners and fish farmers in Malawi and cited poor feeding, among other management practices, as the cause of the poor survival rates recorded. This represents the biggest challenge to small scale farmers, a category to which most of the local farmers belong.
Matching the type of feed to the ontogeny of the alimentary tract of the fish is crucial to fish survival (Hecht, 2013). Exogenous feeding in fry starts immediately after the exhaustion of yolk reserves. However, it has been noted that at this stage, the gastro-intestinal tract of African catfish fry is not fully developed to effectively utilize dry feeds as is the case in tilapine fry (Viveen, 1984; Hecht, 2013). Only after the stomach becomes functional and pepsin activity contributes significantly to protein digestion can the larvae be weaned from live food to a dry feed such as Soybean meal (Verreth and van Tongeren, 1989; Verreth et al., 1993). Notably, these (the dry feeds) are the common feeds for fry in Malawi. Consequently, this explains why feeding fry on such feeds results in high mortality unlike in systems where fingering production is based on the use of Artemia salina nauplii diets (Hogendoorn and Visman, 1980; Olurin et al., 2012). The local economic conditions imply Artemia nauplii is largely unavailable and/or unaffordable for small scale farmers and thus the potential of milk sources of protein in improving survival in the species needs to be looked into.
Soymilk and cow milk are the most common milk protein sources in Malawi. Soymilk has a comparable crude protein content and amino acid profile to cow milk (Akinnuli and Olabanji, 2013), is easier to access and is far cheaper. In addition, the use of cow milk in catfish fry nursing would be inconsiderate and unjustifiable in a country where consumption of milk products remains very low, estimated to be 4-6 kg/capita/year (Tebug et al., 2012). This is lower than the African average of 15 kg/capita/year, and far lower than the WHO recommended intake of 200 kg/capita/year (Banda, 2008; Akaichi and Revoredo-Giha, 2014). Therefore, the use of Soybean milk to improve survival rates in African catfish fry would represent a cheaper, socially acceptable and readily available alternative to Artemia for small scale farmers. In addition, De Silva and Phuong (2011) reported the successful use of soymilk among a great majority of striped catfish (Pangasianodon hypophthalmus) nursery farms in Mekong delta, Vietnam. That being said, a common challenge in the use of Soybean meal and other Soybean products in fish feeding is the presence of trypsin inhibitors (Serine Protease inhibitors) in untreated beans which hinder growth. It is also important to determine if such problems exist in Soymilk. Further, the authors are of the view that efforts to improve survival of fry of the species should not hinder growth. Thus, the present study was aimed at assessing both the growth performances and survival rates of African catfish fry fed on Soymilk based diets.
1 Result
1.1 Growth of African catfish fry
African catfish fry in treatment 1 (egg yolk based diet) was stocked at mean weight of 0.027 323 g ± 0.004 66. Fry in treatment 2 (diet based on soymilk prepared from raw soybeans) was stocked at mean weight of 0.026 43 g ± 0.004 334 while fry in treatment 3 (diet based on soymilk prepared from roasted soybeans) was stocked at a mean weight of 0.320 0 ± 0.004 359. ANOVA showed that the stocking sizes of fry in the three treatments did not differ significantly (P = 0.65).
African catfish fry in treatment 1 (egg yolk based diet) had the highest mean final weight (0.657 67 g ± 0.076 912). However, this was found to be not statistically different (P = 0.8) to the mean final weight of fry in treatment 3 (diet based on soymilk prepared roasted soybeans) (0.611 10 g ± 0.034 66). The mean final weight of fry was found to be lowest in treatment 2 (diet based on soymilk prepared raw soybeans) (0.238 62 g ± 0.028 185) and this was significantly different from fry fed on diets based on egg yolk diet and soymilk from roasted soybean (P = 0.0028 and 0.0051, respectively) as presented in Table 1.
|
Table 1 Initial and final mean weights, Average Daily Weight Gain (ADWG), increase in body weight and specific growth rate (SGR) of fry fed on soymilk and egg yolk based diet |
1.2 Survival rates of African catfish fry
The results of the experiment showed that survival of catfish fry is highest in fry fed on a diet based on soymilk from roasted beans (treatment 3). This was however not statistically different to survival rate of catfish fed on a diet based on soymilk from raw soybeans (treatment 2). Lowest survival was recorded in catfish fry fed on egg yolk based diet (treatment 1). This was statistically lower than fry in treatments 2 and 3. Figure 1 below summarizes the survival rate of African catfish fry after 42 days.
|
Figure 1 Final survival rate (%) of African catfish fry after 42 days of feeding |
2 Discussions
2.1 Growth of African catfish fry
The lack of statistically significant differences between fry in treatment 1 (egg yolk based diet) and fry in treatment 3 (diet based on soymilk from roasted soybeans) as shown in Table 1 was surprising because egg yolk, an animal protein source, has a superior amino acid profile to soybeans and fry fed on egg yolk based diets would thus be expected to show superior growth. These results obtained can be explained to some extent by the observations by Chow (1980) who explored ramifications of feeding eggs to fish larvae.
Egg yolk is highly nutritious. However, it has a very high energy-protein ratio which could result in inadequate intake of protein necessary for maximum growth when used as a diet for very young fish. This is because animals, fish not exempted, eat to satisfy their energy requirements. A fish's food intake increases or decreases corresponding to a reduction or an increase in the diet's energy density. For diets of fixed protein composition, the protein requirement may not be sufficiently met if the diet has too high an energy-protein ratio resulting in increased lipid deposition and reduced growth (Chow, 1980). This concurs with observations by Takeda et al., (1975), Wanatabe (1982) and Daniels and Robinson, (1986).
The optimum dietary energy-protein for fish differs among species and at various stages of the life history. Hosain et al., (1998) recommends an energy-protein ratio of 122 for diets of young African catfish, which was also found to be optimal for Channel catfish fry (Ictalurus punctatus) by Winfree and Stickney (1984). A normal chicken egg yolk has 32-33% CP and metabolisable energy content of around 5 700 kcal/kg (Chow, 1980; Bologa et al., 2013), thus an energy-protein ratio of between 173 and 178 which is considerably over the recommended ratio. This, and the inclusion of fishmeal in both treatments after 1 week, helps to explain why growth of fry fed on egg yolk was not significantly better than growth of fry whose diet was based on soymilk from roasted beans despite the egg yolk having a superior nutritional profile. Similar observations on the effect of diets with very high energy-protein ratios on growth of young fish were made in hybrid striped bass Morone chrysops X M. saxatilis (Nematipour et al., 1992). Marammazi and Kahkesh (2011) also documented reduced growth of juvenile Shirbot (Barbus grypus) fed on diets with high energy-protein ratios.
Observations from the present study also show that growth of catfish fry was poor in fry fed on a diet based on soymilk from raw (unroasted) beans. We attribute this to the presence of anti-nutritional factors that are presented in untreated soybeans such as trypsin inhibitors and phytic acid. It has been documented that roasting or any other form of heat treatment helps to deactivate these inhibitors and improve protein utilization by fish. During the roasting process, active Kunitz soybean trypsin inhibitor decreases from 1.8% of raw soy flour protein to only 0.57% soy flour proteins while the Bowman-Birk inhibitor family comprises 0.38% and only 0.04% of raw and roasted soy flour respectively (Anderson, 1986). Ogbonna et al., (2014) found that growth of African catfish fingerling was significantly better in fingerlings fed on a roasted soybean meal diet than fingerlings fed on a diet of raw soybean meal and similarly attributed the differences to the presence of anti-nutritional factors in raw soybean.
Results from the study clearly demonstrate that diets based on soymilk from roasted beans can be used to feed African catfish fry as growth of the fry was highest in fry fed on the diet, alongside fry fed on egg yolk diet, and was far superior to the growth of fry fed on diets based on soymilk prepared from raw soybeans.
2.2 Survival rates of African catfish fry
Several studies have documented that live food in the form of enriched Artemia nauplii, small Daphnia, Moina or other zooplankton of suitable size are essential for survival and is preferred to dry feed, in this case boiled egg yolks (Hecht, 2013). This is because the stomach of the young fish is not fully functional at the start of exogenous feeding. Therefore, high survival rates can only be obtained if feed provided matches the development of the gut of the fish.
Results from our experiment demonstrate that providing feed in a ‘milk’ or filtrate form as was the case with the soymilk has the same effect on the digestive tract. That is to say, soymilk matches with the ontogeny of the digestive system of the fish. This is supported by observations on survival rate of fry in our study which showed that survival rate of fish in this experiment was significantly higher in fry fed on diets based on soymilk prepared from raw soybeans and soymilk from roasted soybeans (treatments 2 and 3). Survival of fry in both treatments was significantly higher than survival of fry fed on a diet based on egg yolk diet (treatment 1). de Graaf and Jensen (1996) obtained survival rates of 40% over a 50-day experimental period following what was described as ‘good feeding and management practices’. This is consistent with the results of the present study and supports our conclusion that soymilk is ideal for African catfish fry nursing to improve survival.
The differences in survival of fry fed on diets based on soymilk and those fed on diets based on egg yolk can also be attributed to differences in cannibalistic activities in the treatments. During the course of the experiment, a total of 18 ‘jumper’ fry were isolated and graded from tanks in which fry were fed on egg yolk based diet. In contrast, only 4 were graded from fry fed on diets based on soymilk prepared roasted soybeans and none from fry fed on diets based on soymilk prepared from raw soybeans. This implies that cannibalism was greatest in fry fed on diets based on egg yolk.
The differences in cannibalistic activity were more likely a result of the differences in the form in which the feed was presented than the type of the feed itself. Almazán et al., (2004) showed that a reduction in swimming activity results in a lower incidence of aggressive behavior. Hecht (2013) suggests that feeding frequency, food availability influences swimming activity of fry and Broom (1998) demonstrated that aggression and other behavior patterns can be altered by feed availability. The soymilk was presented in form of a filtrate that was dispersed evenly into the rearing units. The egg yolk, on the other hand, was presented in form of crumbs that were likely not evenly distributed in the tanks. This caused intra-tank differences in feed availability causing an increase in swimming activity in search of feed and the resulting aggression. This also likely resulted in some individual fry having better access to the feed than others (the jumpers). This, in turn, explains the high cannibalism observed.
From this study, it is evident that diet of African catfish fry based on soymilk prepared from roasted soybeans is best for obtaining high survival rate of the fry as in both treatments where soymilk was used to feed fry, survival rate was statistically higher than in the treatment where egg yolk was instead used to feed fry among fry in treatment 1. While the study has demonstrated positive effects of diets based on soymilk from roasted beans on growth and shows higher survival rates of African catfish fry than previous studies in the country (Iita, 2013), the study did not vary the application rate (or feeding rate) of the soymilk. Therefore, it is difficult to assert that the application rate used in this study (1 ml of soymilk / litre of culture media) is suitable for optimal growth and survival of African catfish fry. That is to say, it is possible that the application rate used in this study is below or above some hypothetical optimal application rate that can be established through further research by investigating growth and survival response of fry when soymilk application rate is varied.
2.3 Water Quality
Temperature values were within recommended limits for African catfish larvae and fry growth and survival. Barton et al., (2002) recommends that temperatures should be within 23oC - 30oC agreeing with FAO (1996). The study was limited by a lack of automated water heaters at the experiment to regulate water temperature resulting in mean temperatures that were below the optimum 28oC for growth of African catfish as reported by Hecht (2013) as presented in Table 2. Fish growth decreases at temperatures above or below the optimum temperature (Stickney, 1994), and this suggests possible higher growth rates of fry than presently reported in this study, if temperature was regulated.
|
Table 2 Water quality parameters (mean ± SE) measured during the experimental period |
pH values recorded in all treatments were within recommended limits during the duration of the experiment as they were within the recommended range of 6.0-9.0 while DO levels were similarly within optimal levels as they were above 6 mg/l in all treatments (FAO, 1996).
Ammonia concentration in all treatments was however found to be above the recommended limit of 0.05 mg/l for optimal growth and survival (Pillay, 1990). Ammonia concentration in treatments 2 and 3 (where soymilk was fed to fry) was significantly higher than treatment 1 (egg yolk based diet). This is somewhat expected as in both treatments, the protein rich soymilk was directly released into the culture medium. This resulted in high concentration of ammonia as ammonia is a waste product of protein. However, the ammonia concentrations in all treatments were below the maximum tolerable limit of 1 ppm (1 mg/L). Thus, the study shows that if a proper water quality management regime is followed, as the one followed in this study, it is possible to keep ammonia levels within tolerable levels. The study also showed that application of soymilk in rearing tanks has no effect on other water quality parameters that were monitored during the experiment.
3 Materials and Methods
The experiment was conducted at the Bunda fish farm of The Lilongwe University of Agriculture and Natural Resources located 2 Km from the main campus. The campus is located 32.5 Km south of Lilongwe city in the central region of Malawi. The site was chosen due to availability of technical support and materials that were required during the course of the study (Figure 2).
|
Figure 2 Study area (Source: http://www.maplandia.com/Malawi/Lilongwe) Note: Satellite view of Bunda Fish Farm, the study site |
3.1 Experimental Fish
Clarias gariepinus larvae were obtained by semi-artificial propagation of selected brood stock at the Bunda fish farm hatchery using the method described by de Graaf and Jensen (1996). Brood stock in ponds at the farm were inspected for readiness to breed and six brooders (2 females and 4 males) with body weights between 265-350 g were selected for breeding. The selected fish was then injected with Aquaspawn (Spawnrite Ltd) at a full dose of 0.5 ml/Kg for the females and half dose for the males (0.25 ml/Kg). The brooders were then confined in a glass fiber tank with synthetic substrates and an air stone aerator at a sex ratio of 2:1 (males: females) to promote courtship and spawning overnight. Brood fish was then removed from the tank the next morning following successful spawning. Eggs attached to the substrate were incubated at a temperature of 27.8oC.
Hatchlings (larvae) were reared inside the hatchery for a period of 7 days. During this period, Absorption of yolk reserves was monitored under a microscope and found to be completely depleted after 3 days and this coincided with anal opening. At this stage, the larvae was then said to have graduated to fry and was fed on fresh cattle blood and planktons fed till satiation, 5 times per day for a period of 4 days to stimulate digestive activity before being randomly allocated to fiberglass tanks outside the hatchery at a stocking density of 500 fish/tank with water volume of 500 litres. Growth measurements and survival observations were taken for a period of 42 days.
3.2 Experimental Design and Treatments
The experiment was laid out in a Completely Randomized Design (CRD). The experiment had three treatments. These were;
Ø Treatment 1: control; Fry fed on boiled egg yolk + fish meal + fresh water planktons. This was chosen as the control because it is a common diet for African catfish fry at Bunda fish farm.
Ø Treatment 2: Fry fed on soymilk prepared from raw soybeans + fish meal + fresh water planktons
Ø Treatment 3: Fry fed on soymilk prepared from roasted soybeans + fish meal + fresh water planktons
Each treatment was replicated three times.
3.3 Experimental feeds and feeding
Eggs, from which the yolk was extracted to feed the fry, were bought from the Bunda farm shop. The eggs were soft boiled and the shells removed. Thereafter, the white part of the boiled eggs (albumen) was removed as well so that only the yolks remained. The egg yolks were broken down into small crumbs and 100 ml of water was added so that the yolk was suspended in water. Soybeans, from which Soymilk was prepared, were bought from Mitundu market in Lilongwe district, central Malawi. One half of the purchased soybeans, equivalent to 10 kg, was roasted till the colour turned dark brown and cracks appeared before milling for soymeal. This was done to deactivate trypsin inhibitors. Figure 3 outlines the process of preparation of soymilk used in the experiment.
|
Figure 3 Steps followed during the preparation of soymilk for catfish fry feeding |
The fish was fed 5 times every day, between 8 am and 4 pm, at 2 hour intervals, following recommendations by Nagoli and Balasubramaniam (2008) and Hecht (2013). During the first week of the experiment, fry in treatment 1 was fed on an egg yolk diet with plankton supplements while fry in treatment 2 and 3 was fed on a diet of soy milk prepared from unroasted and roasted beans respectively, and plankton supplements. Fry fed on egg yolk diet was fed till satiation while soy milk was applied in respective tanks at an application rate of 1 ml/liter of water each day. Thus, 500 ml of soy milk was applied in each tank each day.
From the second week of the experiments, fish meal was included in the diets of fry in all treatments. As with the soymeal used in preparing soymilk, the fish meal was double sieved to remove hard residues that were likely to have come from scales and other hard parts of the fish. The fish meal was mixed with water before feeding the fry. The fish meal, fed till satiation, was given to the fry twice each day, at 10 am and 2 pm.
3.4 Data Collection and Analysis
Data on growth, expressed as weight gains of fry, was collected bi-weekly over a period of 6 weeks (42 days) by sampling 50 fry from each tank and recording the weight on an electronic balance each time sampling was done. Survival of experimental fish in each tank was recorded at the end of the experiment by physically counting the number of fry alive. A thumb counter was used to assist the counting process.
Growth rate in each treatment was measured as specific growth rate and average daily weight gain (ADWG). Specific growth rate, average daily weight gain and survival rate was calculated using the following formulae (Pillay, 1990);
i. Average Daily Growth (ADG)
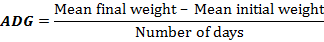
ii. Specific Growth Rate (SGR percentage)
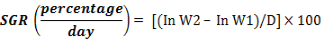
Where: W2= Final weight in grams
W1= Initial weight in grams
D = time interval (number of experimental days)
iii. Body weight gain (BWG)
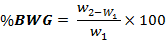
Where, W1 = initial mean weight, W2 = final mean weight
3.5 Water Quality Monitoring and Management
Ammonia level in rearing units was checked bi-weekly using the Phenate method. Temperature, pH and Dissolved oxygen (DO) of water in each tank was recorded twice every day, at 8 am and 2 pm, using a digital water checker.
To maintain good water quality in the rearing tanks, partial water replacement was done by flashing out 10 cm in each tank at the end of each day. Waste feed and faecal matter remaining at the bottom of the tanks was siphoned out after every 3 days. Lost water after siphoning out waste material was replaced. Aeration stones were used to supply oxygen.
Data analysis was done using SPSS (Statistical Package for Social Scientists) Version 20. One-way Analysis of Variance (ANOVA) was used to test for significant differences among treatment means. Tukey HSD test was used to separate means after ANOVA showed significant difference among treatment means.
Authors’ Contributions
DK conceptualized the study and was responsible for coordination among the authors during the design phase of the study. He was also responsible for sourcing materials such as brood stock and tanks that were used during the study. APC and VJMC were responsible for induced propagation of brood stock, management of fry during the experiment including feeding and water exchange, and data collection. All three authors took part in data analysis and drafting of this manuscript.
Acknowledgements
We express our deepest gratitude to Dr. Daniel Sikawa, Mr Robert Ssenfuma, Mr Wei Ying and Mrs Mabohlokoa Khoaisanyane for the support they rendered to us during the course of our experiment. We are also extremely thankful to Mr. Isaacs Chaggwa, the Bunda fish farm manager, and his team for providing us with tanks and other necessary materials to make our experiment possible.
Abdelhamid A.M., Radwan I.A, Mehrim A.I., and Abdelhamid A.F.B., 2010, Improving the survival rate of African catfish, journal of animal and poultry production, Mansoura University., Vol. 1 (9): 409 – 416
Akaichi F. and Revoredo-Giha C., 2014, The demand for dairy products in Malawi, African Journal of Agricultural and Resource Economics, Volume 9, Number 3, pages 214-225
Akinnuli B.O. and Olabanji M.O., 2013, Development and performance evaluation of soya beans milk extracting machine, International Journal of Engineering Science and Innovative Technology (IJESIT), Volume 2, Issue 3
Aquaculture Development in Malawi (ADiM)., 2005, ADiM Working Paper, No. 2, Situation analysis of aquaculture in Malawi, Master Plan Study on Aquaculture Development in Malawi, ADiM, Malawi
Almazán R.P., Schrama J.W., and Verreth J.A.J., 2004, Behavioural responses under different feeding methods and light regimes of the African catfish (Clarias gariepinus) juveniles, Aquaculture, 231: 347–359
https://doi.org/10.1016/j.aquaculture.2003.11.016
Anderson R.L., 1986, Trypsin inhibitors of raw and heat-treated soybean meals (anhydrotrypsin, affinity chromatography), University of Nebraska-Lincoln, USA
Banda J.W., 2008, Revolutionising the livestock industry in Malawi, The 12th University of Malawi Inaugural Lecture, Bunda College, University of Malawi, Lilongwe, Malawi
Barton B.A., Morgan J.D., and Vijayan M.M., 2002, Physiological and condition-related indicators of environmental stress in fish, In: Biological Indicators of Aquatic Ecosystem Health, Adams S.M. (ed.), American Fisheries Society, Bethesda, M.D. pp. 111-148
Bologa M., Pop I.M., and Albu A., 2013, Research on chemical composition of chicken egg from different system of production (conventional and organic), Lucrări. Şti. Zoot., 59, 80–85
Broom D.M., 1998, Fish welfare and the public perception on farmed fish, Advances in the Study of Behavior, 27: 371-403
https://doi.org/10.1016/S0065-3454(08)60369-1
Chow K.W., 1980, Microencapsulated egg diets for fish larvae, Training Course in Fish Feed Technology, United Nations Development Programme & Food and Agriculture Organization of the United Nations, Rome, Italy, pp. 24-31
Daniels W.H. and Robinson E.H., 1986, Protein and energy requirements of juvenile red drum (Sciaenops ocellatus), Aquaculture, 73: 243-252
https://doi.org/10.1016/0044-8486(86)90354-6
de Graaf G. and Jensen J., 1996, Handbook on the artificial reproduction and pond rearing of the African catfish claris gariepinus in sub-Saharan Africa, FAO Fisheries Technical paper 362, NEFISCO foundation, Amsterdam, the Netherlands
De Silva S.S. and Phuong N.T., 2011, Striped catfish farming in the Mekong Delta, Vietnam: a tumultuous path to a global success, Reviews in Aquaculture, (2011) 3: 45–73
https://doi.org/10.1111/j.1753-5131.2011.01046.x
Ecker O. and Qaim M., 2011, Analyzing nutritional impacts of policies: an empirical study for Malawi, World Development, 29(3): 421-428
https://doi.org/10.1016/j.worlddev.2010.08.002
FAO, 1996, Fisheries and food security, Documentation issued on the occasion of the world food summit in Rome (1996), Food and Agriculture organization of the United Nations, Rome, Italy 2pp
FAO, 2006, Food and agriculture organization of the united nations, Rome, Italy, Aquaculture production, 2005, Year book of Fishery Statistics - Vol.96/2
Goos H.J. and Richter C.J.J., 1996, Internal and external factors controlling reproduction in African catfish, Clarias Gariepinus, Journal of Aquatic Living Resources, Vol. 9 45-58
https://doi.org/10.1051/alr:1996041
Hecht T., 2013, A review of on-farm feed management practices for North African catfish (Clarias gariepinus) in sub-Saharan Africa, In: Hasan M.R., and New M.B. (ed.), On-farm Feeding and Feed Management in Aquaculture, FAO Fisheries and Aquaculture Technical Paper No. 583, Rome, Italy, pp.463–479
Hogendoorn H. and Vismans M.M., 1980, Controlled propagation of the African catfish, Clarias lazera (C&V) and Artificial reproduction, Aquaculture, 21: 39-53
https://doi.org/10.1016/0044-8486(80)90124-6
https://doi.org/10.1016/0044-8486(80)90133-7
Hosain M.A., Begum S., Islam M.N. and Shah I.K.M.A., 1998, Studies on the optimum protein to energy ratio of African catfish (Clarias gariepinus Burchell), Bangladesh Journal of Fisheries Research, 2 (1), 1998: 47-54
Iita T.I., 2013, Effects of feeding regimes, rearing units on the survival and growth of African catfish Clarias gariepinus (Burchell 1822) larvae and the reproductive performance, Thesis for M.S., Bunda College of Agriculture, University of Malawi, Supervisor: Kassam D., pp. 3-5
Mammarazi J.G. and Kahkesh F., 2011, Effects of dietary protein and energy levels on growth perfomance, feed utilization and body composition of juvenile shirbot barbus grypus (Heckle, 1843), Iranian Journal of Fisheries Science, 10(3) 461-474, 2011
M’balaka M., Kassam D., and Rusuwa B., 2013, Effect of stocking density on the growth and survival of improved (F5 and F6) and unimproved native strains of oreochromis shiranus (trewavas) raised in hapas, Indian Journal of Fisheries, 60(2) :151-155, 2013
Nagoli J. and Balasubramaniam A., 2008, Natural Spawning of the African catfish (Clarias gariepinus), The World Fish Center, Zomba, Malawi
Nematipor G.R., Brown M.L., and Gatlin III D.M., 1992, Effects of dietary energy: protein ratio on growth characteristics and body composition of hybrid striped bass, Morone chrysops X M. saxatilis, Aquaculture, 107 (1992) 359-368
https://doi.org/10.1016/0044-8486(92)90083-W
Njaya F., 2015, Ecosystem approach to fisheries and aquaculture for southern Lake Malawi and Lake Malombe: rebuilding chambo stocks for increased socio-economic benefits of the local community, Department of Fisheries, Lilongwe, Malawi
Nyina-Wamwiza L., Wathelet B., and Kestemont P., 2007, Potential of local agricultural by-products for the rearing of African catfish Clarias gariepinus in Rwanda: effects on growth, feed utilization and body composition, Aquaculture Research, vol. 38, no. 2, pp206–214
https://doi.org/10.1111/j.1365-2109.2007.01658.x
Ogbonna S.U., Uhuo C.A., Okereke C.N., Azi S.O., and Nwanchor K.C., 2014, Effects of Soybean meal (Glycine max) on the Growth Performance and Feeding Habits of Clarias gariepinus fingerlings Reared Under Laboratory Condition, Sky Journal of Biochemistry Research Vol. 3(3), pp. 033 – 036
Olurin K.B., Iwuchukwu P.O., and Oladapo O., 2012, Larval rearing of African catfish, Clarias gariepinus fed decapsulated Artemia, wild copepods or commercial starter diet, African Journal of Food Science and Technology (ISSN: 2141-5455), Vol. 3(8), pp.182-185
Pillay T.V.R., 1990, Aquaculture principles and practices, Fishing Newsbooks, Rome, Italy
Rad F., Kurt G.I., and Bozaoủlu A.S., 2003, Effects of spatially localized and dispersed patterns of feed distribution on the growth, size dispersion and feed conversion ratio of the African catfish (Clarias gariepinus), Turkish Journal of Veterinary Animal Science, 28, 851-856
Stickney R.R., 1994, Principles of aquaculture, John wiley and Sons, New York, USA
Takeda M., Shimeno S., Hosokawa H., Kajiyama H., and Kaisyo T., 1975, The effect of dietary calorie to protein ratio on growth, feed conversion and body composition of young yellowtail, Bulletin of the Japanese Society for the Science of Fish., 41: 443-447
https://doi.org/10.2331/suisan.41.443
Tebug S.F., Chikagwa-Malunga S., and Wiedemann S., 2012, On-farm evaluation of dairy farming innovations uptake in northern Malawi, Livestock Research for Rural Development 24(5): 1–9The United States Agency for International Development (USAID)., 2015, The Importance of Wild Fisheries for Local Food Security: MALAWI, Washington, D.C., United States, Available at: https://rmportal.net/biodiversityconservation-gateway/resources/projects/measuring-impact/mi-project-resources/the-importance-of-wild-fisheries-for-local-food-security/the importance-of-wild-fisheries-for-local-food- security-malawi/view [Accessed on 27 June, 2016]
Verreth J. and Van Tongeren M., 1989, Weaning time in Clarias gariepinus (Burchell) larvae, Aquaculture, 83: 81–88
https://doi.org/10.1016/0044-8486(89)90062-8
Verreth J., Eding E.H., Rao G.R.M., Huskens F., and Segner H., 1993, A review of feeding practices, growth and nutritional physiology in larvae of the catfishes Clarias gariepinus and Clarias batrachus, Journal of the World Aquaculture Society, 24: 135–144
https://doi.org/10.1111/j.1749-7345.1993.tb00002.x
Viveen W.J.A.R., Richter C.J.J., Van Oordt P.G.W.J., Janssen J.A.L., and Huisman E.A., 1985, Practical manual for the culture of the African catfish (Clarias gariepinus), The Netherlands Ministry for Development Cooperation, Section for Research and Technology, P.O. Box 20061, 2500 EB The Hague, The Netherlands, 128 pp
Wanatabe T., 1982, Lipid nutrition in fish, Comparative Biochemistry and Physiology, 73: 3–15
Winfree R.A. and Stickney R.R., 1984, Starter diets for channel catfish: Effects of dietary protein on growth and carcass composition, Progress in Fish Culture, 46(2): 79-86
. PDF(438KB)
. FPDF(win)
. HTML
. Online fPDF
Associated material
. Readers' comments
Other articles by authors
. A.P. Chelewani
. D. Kassam
. V.J.M. Chiwanda
Related articles
. African catfish
. Clarias gariepinus
. Soymilk-based
. Fry stage
. Growth rate
. Survival rate
Tools
. Email to a friend
. Post a comment
.png)
.png)
.png)
.png)
.png)


